Reactivity chart chemistry12/3/2023 This allows for electrons to be retained as a result of these attractive forces. An example of high ionization energy is in helium, as it has a very small atomic radius with +2 charge present in the nucleus. The Ionization energy increases further right down the period, and higher up a group as more energy is required to remove a tightly bounded electron from the atom. This property is describing the difficulty of removing an electron. The minimum amount of energy required to remove an electron from a neutral atom’s outermost electron shell in the gaseous phase is called the ionization energy. Note: This periodic trend excludes the Noble gases (Group 18). Note that the noble gases do not have an electronegativity. This is observed through Periodic Trends, as the further right of a period, and higher up a group are observed to have increased values in electronegativity. This scale ranges from 0.7 to 4.0, where cesium is the least electronegative element, and with fluorine being the most electronegative element. It was created by measuring the bond energy of the different elements joined by covalent bonding. The scale that was formed in order to measure this property is the Pauling scale. 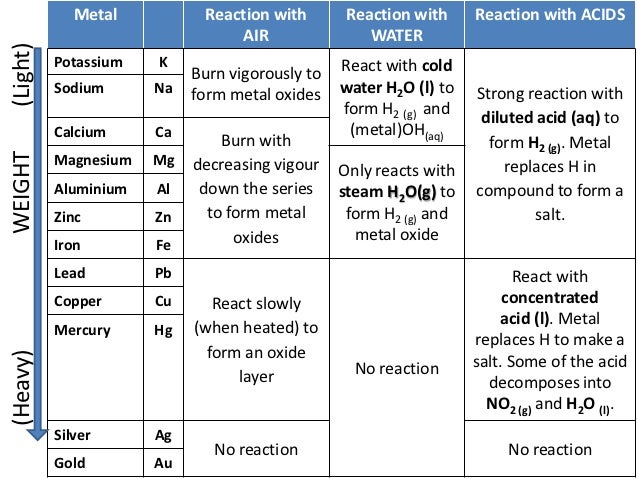
Which properties can be identified using periodic trends? Electronegativity:Įlectronegativity is a property that measures the tendency of an atom to attract electrons to form a bond. We discuss trends for properties such as electronegativity, atomic radius, first ionization energy, reactivity and electron affinity. This is due to the structural similarities’ elements have within a period or family that allows for these trends to take place. These trends have allowed scientists in the past to predict certain characteristics of unknown elements. Periodic trends are observable patterns in the properties of an element that are dependent on its position in the Periodic Table. The following chart summarizes the reactions discussed above. Iron will react with sulfuric acid to give hydrogen and iron (II) sulfate.įe (s) + H 2SO 4 (l) → FeSO 4 (aq.) + H 2 (g) Zinc (Zn) reacts with sulfuric acid (H 2SO 4) to produce zinc sulfate (ZnSO 4) while liberating hydrogen gas (H 2). Metals that lose electrons easily produce a fast and vigorous reaction.ġ. The reactivity of the metal determines the reaction rate. The amount of hydrogen produced depends on the starting mass of the metal and the acid’s strength. The electrons and protons combine to form a hydrogen atom. Each metal atom loses one or more electrons, and each acid molecule loses one or more protons. Highly reactive metals produce hydrogen when reacting with acid. Aluminum (Al) reacts vigorously with water to produce hydrogen gas.Ģ Al (s) + 6 H 2O (l) → 2 Al(OH) 3 (aq.) + 3 H 2 (g) 3. Sodium (Na) is highly reactive and displaces hydrogen (H 2) from water (H 2O).Ģ Na (s) + 2 H 2O (l) → 2 NaOH (aq.) + H 2 (g)Ģ. Highly reactive metals can react with cold water to form the corresponding hydroxide.ġ. Magnesium (Mg) can displace copper (Cu) in an aqueous solution of copper nitrate (Cu(NO 3) 2). Copper (Cu) can displace silver (Ag) in an aqueous solution of silver nitrate (AgNO 3).Ĭu (s) + 2 AgNO 3 (aq.) → Cu(NO 3) 2 (aq.) + 2 Ag (s/ppt.)Ģ. A characteristic of this reaction is that one cation trades places with another to form a new product.ġ. This reaction is known as a single-displacement reaction. Metals higher in the series tend to lose electrons and easily displace lower metals. Based on the importance of the reactivity series, let was look at the various chemical reactions that metals undergo. It helps us understand why different metals are extracted from their ores differently. Aside, it can predict the outcome of single-displacement reactions. It can predict the reactions between metals and water and metals and acids. The reactivity series is essential for several reasons. įrom this list, it is clear that potassium is the most reactive metal. The list below represents the order of the reactivity series from most reactive to least reactive. All metals above hydrogen liberate hydrogen gas (H 2) upon reacting with dilute hydrochloric acid (HCl) or dilute sulfuric acid (H 2SO 4).Metals at the bottom of the chart, gold, silver, and platinum, usually are unreactive and do not react with acid and water.The reducing ability of metals decreases as we go down the series.They are also strong reducing agents and oxidize quickly. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
